Targeted creation of new mutants with compact plant architecture using CRISPR/Cas9 genome editing by an optimized genetic transformation procedure in cucurbit plants
Citation: Tongxu Xin, Haojie Tian, Yalin Ma, Shenhao Wang, Li Yang, Xutong Li, Mengzhuo Zhang, Chen Chen, Huaisong Wang, Haizhen Li, Jieting Xu, Sanwen Huang, Xueyong Yang, Targeted creation of new mutants with compact plant architecture using CRISPR/Cas9 genome editing by an optimized genetic transformation procedure in cucurbit plants, Horticulture Research, Volume 9, 2022, uhab086, https://doi.org/10.1093/hr/uhab086
Horticulture Research
Published: 20 January 2022
Abstract
Fruits and vegetables in the Cucurbitaceae family contribute greatly to the human diet, for example, cucumber, melon, watermelon and squash. The widespread use of genome editing technologies has greatly accelerated the functional characterization of genes as well as crop improvement. However, most economically important cucurbit plants, including melon and squash, remain recalcitrant to standard Agrobacterium tumefaciens-mediated transformation, which limits the effective use of genome editing technology. In this study, we describe the “optimal infiltration intensity” strategy to establish an efficient genetic transformation system for melon and squash. We harnessed the power of this method to target homologs of the ERECTA family of receptor kinase genes and created alleles resulting in a compact plant architecture with shorter internodes in melon, squash and cucumber. The optimized transformation method presented here allows stable CRISPR/Cas9-mediated mutagenesis and will lay a solid foundation for functional gene manipulation in cucurbit crops.
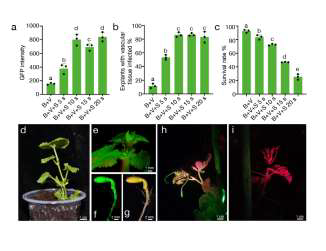
The transgenic T1 plant (h) and WT (i) of melon under the GFP channel of LUYOR-3415RG Dual Fluorescent Protein Lamp. Tissues appear green due to GFP expression and red due to the autofluorescence of chlorophyll.
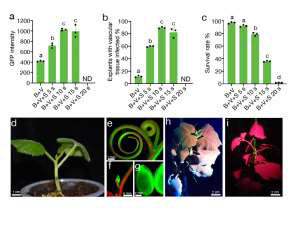
(d) A regenerated transgenic T0 plant of squash after one week of transferring to soil. The GFP fluorescent tendril (e), apical meristem(f) and seeds (g) of the transgenic squash T0 plant. The transgenic T1 plant (h) and WT (i) of squash und GFP channel of LUYOR-3415RG Dual Fluorescent Protein Lamp.
LUYOR-3415RG Dual Fluorescent Protein Lamp: http://www.luyorgroup.com/Fluorescent-Protein-light-sources/LUYOR-3415RG.html